BioSocial Health J. 1(2):74-83.
doi: 10.34172/bshj.22
Systematic Review
The effects of artificial sweeteners on body weight, body fat, and energy intake: A meta-analysis of meta-analyses
Fatemeh Hamedi-Kalajahi Investigation, Writing – original draft, 1 
Sanaz Asemani Conceptualization, Data curation, Investigation, Methodology, Visualization, Writing – original draft, Writing – review & editing, 1 
Kousalya Prabahar Investigation, Methodology, Writing – original draft, 2
Neda Jourabchi-Ghadim Conceptualization, Data curation, Formal analysis, Investigation, Software, Visualization, Writing – original draft, 1
Alireza Ostadrahimi Conceptualization, Data curation, Investigation, Project administration, Supervision, Validation, Visualization, Writing – original draft, 3, 4, * 
Author information:
1Student Research Committee, Tabriz University of Medical Sciences, Tabriz, Iran
2Department of Pharmacy Practice, Faculty of Pharmacy, University of Tabuk, Kingdom of Saudi Arabia
3Nutrition Research Center, Tabriz University of Medical Sciences, Tabriz, Iran
4Department of Clinical Nutrition, Faculty of Nutrition and Food Science, Tabriz University of Medical Sciences, Tabriz, Iran
Abstract
Introduction:
Overweight and obesity are prevalent worldwide and hence it can be considered as a global epidemic. Artificial sweeteners (ASs) are suggested to decrease energy intake and prevent obesity, however, the results of meta-analyses are inconsistent. Therefore, this umbrella meta-analyses was conducted to resolve these discrepancies and offer definitive evidence on the impact of ASs on body weight (BW), body mass index (BMI), and energy intake.
Methods:
PubMed, Scopus, EMBASE, and Web of Science were systematically searched from inception up to March 2023. Meta-analyses studies investigating the effect/association of AS on/with BW, BMI, and energy intake were included. Random-effects model was used for performing a meta-analysis. Subgroup analyses were carried out based on various independent variables. The quality of the included meta-analyses was evaluated using the AMSTAR2 questionnaire.
Results:
In this umbrella review, 12 meta-analyses were included. The findings from the umbrella meta-analysis of interventional studies showed that AS consumption significantly reduced BW according to standardized mean difference (SMD) analysis (ES=-0.38; 95% CI: -0.56, -0.20, P≤0.001), but not when using the weighted mean difference (WMD) (ES=-0.45; 95% CI: -1.15, 0.24, P=0.20). Also, AS did not affect the energy intake (ES=-0.28; 95% CI: -1.15, 0.59, P=0.59) based on WMD. However, AS significantly reduced BMI (ES=-0.28; 95% CI: -0.40, -0.15, P≤0.001) based on WMD analysis. Conversely, the umbrella meta-analysis of observational studies found that consuming ASs was associated with a 61% augmented risk of obesity and overweight. (ES=1.61; 95% CI: 1.36,1.87, P≤0.001).
Conclusion:
Despite observational studies, interventional studies show the benefits of ASs consumption. Future studies should be conducted focusing on the dose, types, and formulations of ASs, and more importantly, short-term and long-term consumption of ASs.
Keywords: Sweetening agents, Body weight, Energy intake, Meta-analysis
Copyright and License Information
© 2024 The Author(s).
This is an open access article distributed under the terms of the Creative Commons Attribution License (
http://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Overweight and obesity are prevalent worldwide and hence it can be considered as a global epidemic. Obesity is defined by an excessive augmentation of body fat that negatively impacts an individual’s health.1 It is a significant public health concern contributing to various chronic disorders like cardiovascular diseases and diabetes mellitus.2 Obesity also increases cancer risk.3 There are numerous adverse effects of obesity including depression, anxiety, the feeling of low esteem, and loss of self-confidence.4 Moreover, obesity reduces the life expectancy of people.5 The chronic adverse effects of overweight and obesity affect the economy of an individual and the nation. Direct health care costs play a crucial role in the treatment of obesity and related disorders.6
Consumption of sugar adds fuel to this obesity condition7 and hence there is an increased use of artificial sweeteners (ASs) like sucralose, aspartame, stevia, and saccharin among the public.8 ASs give the satisfaction of eating sugar with no calories. They also can be recommended as a source of reduced sugar intake and hence to reduce body weight (BW).9 Strategies to reduce obesity should concentrate on both decreasing the intake of energy and rising the expenditure of energy. Consumption of low-energy foods is a way of reducing intake of energy and weight loss. Substituting sugar with ASs would also help people in decreasing their energy intake.10
Animal studies have suggested that intake of ASs for a longer duration would result in increased food intake, adiposity, and weight gain.11 Human studies have also reported that ASs may exhibit serious consequences on the metabolism of glucose and control of appetite.12 The Academy of Nutrition and Dietetics proposed that ASs will reduce the intake of energy which in turn helps to reduce BW.13 In contrast, They have been associated with obesity and increased weight.14 A meta-analysis conducted previously provided conflicting results. RCTs reported benefits of ASs in losing weight and observational studies reported a slight increase in body mass index (BMI).15
Clinical practice guidelines have suggested substituting high-calorie sweeteners with ASs to reduce the intake of energy.8 Even though people started to consume ASs in recent times, obesity persists and hence there is a conflict on the part of ASs on obesity. ASs were suggested as an alternative to sugar substitutes and were proposed to be healthier.16 The association between ASs and chronic disorders like hypertension, obesity, and diabetes remains controversial.17 Therefore, a comprehensive understanding of the role of ASs in weight management and energy intake is essential. In adults, ASs when compared with water, leads to decreased BW and energy intake.18 In contrast to this, two meta-analyses have reported that no clear and significant evidence exists between the use of ASs and BW.19,20
Hence this umbrella meta-analysis was conducted to overcome this conflict and provide clear evidence on the effect of ASs on BW, BMI, and energy intake and hence its role in obesity. The objectives were to compare the effect of ASs with sugar, water, and placebo.
Methods
This umbrella meta-analysis was conducted following the guiding principle of the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA), as indicated by reference.21
The search strategy of literature
We searched for relevant articles in international scientific databases including PubMed, Scopus, EMBASE, and Web of Science. The search covered all articles from the inception of each database up to March 2023. The search strategy was devised based on the following MeSH terms and keywords: ((((“Sweetening Agents”[Mesh] OR “Artificially Sweetened Beverages”[Mesh] OR “Non-Nutritive Sweeteners”[Mesh] OR “Stevia”[Mesh] OR “Saccharin”[Mesh] OR “Cyclamates”[Mesh]) OR “Aspartame”[Mesh]) OR (((((((((((“sweetening agent”[Title/Abstract]) OR (“artificial sweetener”[Title/Abstract])) OR (“non-nutritive sweetener”[Title/Abstract])) OR (sweetener[Title/Abstract])) OR (stevia[Title/Abstract])) OR (aspartame[Title/Abstract])) OR (saccharin[Title/Abstract])) OR (cyclamates[Title/Abstract])) OR (sucralose[Title/Abstract])) OR (acesulfame[Title/Abstract])) OR (sweetened[Title/Abstract]))) AND ((((((((((“Obesity”[Mesh]) OR “Obesity, Abdominal”[Mesh]) OR “Obesity, Morbid”[Mesh]) OR ( “Body Weight”[Mesh] OR “Weight Loss”[Mesh] OR “Body Weight Changes”[Mesh] )) OR “Overweight”[Mesh]) OR “Body Mass Index”[Mesh]) OR “Adiposity”[Mesh]) OR “Waist Circumference”[Mesh]) OR “Waist-Hip Ratio”[Mesh]) OR (((((((((((((((obesity[Title/Abstract]) OR (“abdominal obesity”[Title/Abstract])) OR (“morbid obesity”[Title/Abstract])) OR (obes*[Title/Abstract])) OR (weight[Title/Abstract])) OR (“body weight”[Title/Abstract])) OR (“weight loss”[Title/Abstract])) OR (“body weight changes”[Title/Abstract])) OR (overweight[Title/Abstract])) OR (“body mass index”[Title/Abstract])) OR (adiposity[Title/Abstract])) OR (BMI[Title/Abstract])) OR (“waist circumference”[Title/Abstract])) OR (“waist-hip ratio”[Title/Abstract])) OR (WHR[Title/Abstract])))) AND (((meta-analysis[Publication Type]) OR (meta-analysis[Title/Abstract])) OR (meta[Title/Abstract])).
Additionally, the search was limited to articles published in the English language. The wild-card term “*” was employed to enhance the sensitivity of the search strategy.
Inclusion and exclusion criteria
This umbrella meta-analysis included studies that examined the effects of ASs on BW (Weighted mean difference [WMD], Standardized mean difference [SMD]), energy intake (WMD), BMI (WMD) along with their corresponding confidence intervals (CIs). Also, observational studies investigating the association between AS and BW were included. Studies that were excluded from the analysis comprised in vitro, in vivo, and ex vivo studies, case reports, quasi-experimental studies, and low-quality studies. The PICO criteria for the present umbrella meta-analysis were as follows: Population/Patients (P: people who were overweight, obese, diabetic or pre-diabetic, and hypertensive); Intervention (I: ASs); Comparison (C: sucrose, water, placebo); Outcome (O: BW, energy intake, BMI).
Methodological quality assessment
Two reviewers independently assessed the methodological quality of the included articles (SA, KP) using the Assessing the Methodological Quality of Systematic Reviews 2 (AMSTAR2) questionnaire.22 In case of any discrepancies, the senior author (AO) was consulted to arrive at a consensus. The AMSTAR2 questionnaire comprises 16 items that are to be answered with “Yes”, “Partial Yes”, “No”, or “Not a Meta-analysis”. The AMSTAR2 checklist was categorized into “Critically low quality”, “Low quality”, “Moderate quality”, and “High quality”. Meta-analyses with a score of 7 or higher were of high quality.
Study selection and data extraction
Two reviewers screened the articles for eligibility (SA, KP) working independently. Initially, authors were reviewed title and abstract, followed by a thorough evaluation of the full text of relevant articles to determine their eligibility for inclusion in the umbrella meta-analysis. Any discrepancies were resolved through consultation with a third author (AO). From the selected meta-analyses, information on the sample size, publication year, study location, type of ASs,type of comparator (sucrose, water, placebo), duration of intervention, effect sizes (ESs) such as WMD and standardized mean difference (SMD), and corresponding CIs for BW, energy intake, BMI were extracted in an Excel spreadsheet.
Data synthesis and statistical analysis
The pooled ES and its corresponding 95% CI were estimated using random-effects models with the restricted maximum likelihood (REML) method.23 The I2 statistic and Cochrane’s Q-test were utilized to identify heterogeneity. We considered an I2 value greater than 50% or a P value less than 0.1 for the Q-test as indicative of substantial between-study heterogeneity.23 Separate analyses were performed for each measure due to the intrinsic differences between SMD and WMD. Subgroup analyses were conducted to identify possible sources of heterogeneity based on predefined variables such as the number of included studies, intervention duration, study quality, type of comparator, and sweetener. Sensitivity analysis was utilized to determine the impact of individual studies on the overall effect size. Stata, version 16 (Stata Corporation, College Station, TX, US) was used to conduct all statistical analyses. A P value less than 0.05 was considered statistically significant.
Results
Systematic review
The flowchart outlining the process of search is depicted in Figure 1. This meta-analysis comprises a collective of 12 meta-analyses published from 2006 to 2022.15,19,20,24-32 During the screening of titles and abstracts, 454 studies were excluded. In the full-text screen of twenty-five articles, one study was excluded because of investigated pediatrics and children.33 Five studies lacked relevant data, while seven were not aligned with our research objectives. Table 1 displays the included studies’ characteristics.15,19,20,24-32 average age of the study participants ranged from 27 to 66 years. In the meta-analysis of RCTs, the duration of interventions ranged from 5 to 48 weeks. Most of the studies were conducted on people who were overweight, obese, diabetic or pre-diabetic, and hypertensive.19,20,24,25,27,28,32 Some have investigated the effect of sweeteners in different conditions and diseases.15,26,29-31 Additionally, the number of studies included in the analyzed meta-analyses ranged from 2 to 29.
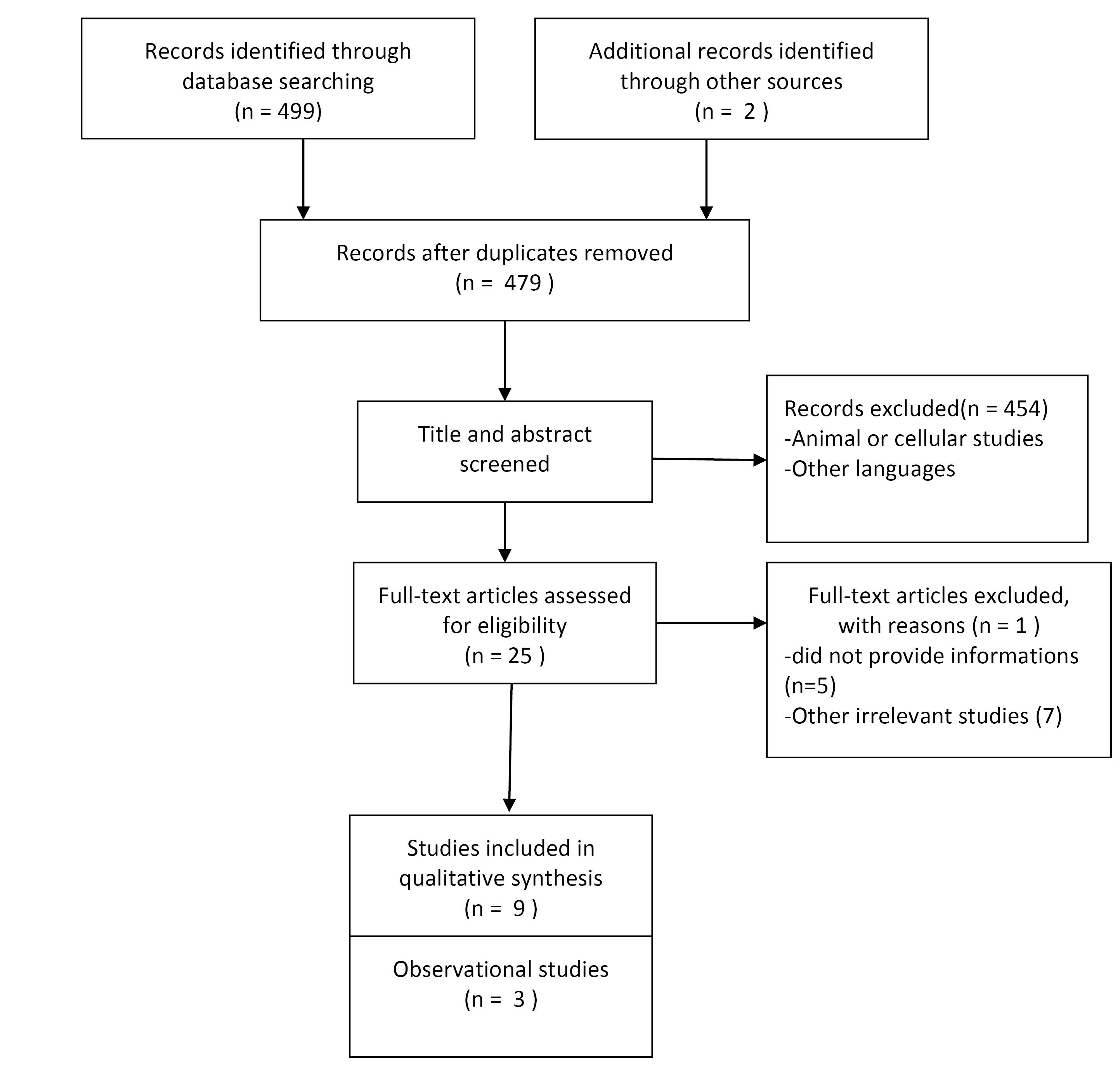
Figure 1.
The study selection process showed by the PRISMA flow chart
.
The study selection process showed by the PRISMA flow chart
Table 1.
Study characteristics of included RCT and Observational studies
Citation
(First author et al, year)
|
No. of
studies in meta-analysis
|
Health condition
|
No. of
participants in
meta-analysis
|
Age (year)
|
Intervention
|
Quality
assessment
scale
|
| Miller and Perez 201415 |
15 |
Different conditions |
1951 |
30 |
Low-calorie sweeteners |
Cochrane Collaboration Risk of Bias tool |
| Azad et al, 201719 |
7 |
Overweight & obese |
1003 |
42 |
Artificially sweetened beverage |
Cochrane Collaboration Risk of Bias tool |
| Toews et al, 2018 20 |
5 |
Healthy & Overweight |
229 |
NR |
Low-calorie sweeteners |
GRADE system |
| Anker et al, 201924 |
7 |
HTN & DM |
460 |
NR |
Stevioside |
Cochrane Collaboration Risk of Bias tool |
| de la Hunty et al, 200625 |
16 |
Healthy & obese |
430 |
NR |
Aspartame |
NR |
| Laviada-Molina et al, 202026 |
20 |
Different conditions |
2999 |
NR |
Low-calorie sweeteners |
GRADE system |
| Lohner et al, 202027 |
8 |
DM2 |
256 |
NR |
Low-calorie sweeteners |
GRADE system |
| McGlynn et al, 202228 |
17 |
Pre-DM and DM |
1733 |
NR |
Low- and no-calorie sweetened beverages |
GRADE system |
| Rogers et al, 202130 |
60 |
Different conditions |
3335 |
NR |
Low-calorie sweeteners |
Cochrane Collaboration Risk of Bias tool |
| Santos et al, 201832 |
12 |
DM and non-DM |
1044 |
45 |
Aspartame |
GRADE system |
|
Observational studies
|
| Azad et al, 201719 |
7 |
Overweight & obese |
1003 |
42 |
Artificially sweetened beverage |
Newcastle–Ottawa Scale |
| Qin et al, 202029 |
5 |
Different conditions |
22390 |
NR |
Artificially sweetened drink |
Newcastle–Ottawa Scale |
| Ruanpeng et al, 201731 |
3 |
Different conditions |
12 987 |
NR |
Artificially sweetened drink |
Newcastle–Ottawa Scale |
Risk of bias assessment
Table 2 provides a summary of the quality assessment results of the meta-analyses using the AMSTAR2 questionnaire.15,19,20,24-32 Nearly all of the meta-analyses included in the umbrella review were assessed as moderate-quality studies. Only one study was assessed as critically low25 and three as high-quality.19,27,30 Among the key domains assessed by AMSTAR2, item 7 was the most frequently neglected in most studies, making it the primary reason for the reduced quality ratings. At this stage, authors should provide a list of excluded studies along with justifications for their exclusion.
Table 2.
Results of assessing the methodological quality of meta-analysis
|
Study
|
Q1
|
Q2
|
Q3
|
Q4
|
Q5
|
Q6
|
Q7
|
Q8
|
Q9
|
Q10
|
Q11
|
Q12
|
Q13
|
Q14
|
Q15
|
Q16
|
Quality assessment
|
| Miller et al, 201415 |
Yes |
Partial Yes |
Yes |
Yes |
No |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Moderate |
| Azad et al, 201719 |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
High |
| Toews et al, 201820 |
Yes |
Partial Yes |
Yes |
Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Moderate |
| Anker et al, 201924 |
Yes |
Partial Yes |
Yes |
Partial Yes |
No |
Yes |
No |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Moderate |
| de la Hunty et al, 200625 |
Yes |
Yes |
Yes |
No |
No |
No |
No |
Yes |
No |
No |
Yes |
Yes |
No |
Yes |
Yes |
No |
Critically low |
| Laviada-Molina et al, 202126 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
No |
Moderate |
| Lohner et al, 202027 |
Yes |
Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
High |
| McGlynn et al, 202228 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Moderate |
| Qin et al, 202029 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
No |
Yes |
Yes |
No |
No |
No |
Yes |
No |
Moderate |
| Rogers et al, 202030 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
High |
| Ruanpeng et al, 201731 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
No |
Yes |
Yes |
No |
Yes |
Yes |
No |
Moderate |
| Santos et al, 201832 |
Yes |
Partial Yes |
Yes |
Partial Yes |
Yes |
Yes |
No |
Yes |
Yes |
No |
Yes |
Yes |
Yes |
Yes |
Yes |
No |
Moderate |
Effect of ASs on BW
Effect of ASs on BW in studies reporting WMD
The impact of ASs on BW was documented in nine meta-analyses that reported WMD. The combined effect sizes of the included studies did not show any significant diminishing in BW (ES = -0.45; 95% CI: -1.15, 0.24, P = 0.20) (Figure 2A). Significant between-study heterogeneity was observed. (I2 = 87.1 %, P ≤ 0.001) (Figure 2A). The intervention duration, study quality, number of included studies, type of comparator, and sweetener could potentially responsible for sources of heterogeneity. In a subgroup analysis of comparator type, AS significantly reduced BW when compared to water. Interestingly, only moderate-quality and low-quality studies demonstrated a significant reduction in BW after AS consumption. Additionally, the effect of AS in diminishing BW was more pronounced in intervention duration of < 10 weeks and in studies with > 5 included studies (Table 3). The sensitivity analysis revealed that excluding one study25 impacted the overall effect size, resulting in a significant reduction in BW due to the effect of ASs (ES = -0.77; 95% CI: -1.15, -0.38). Upon visual examination of the funnel plot (Figure 2B), an asymmetrical distribution of studies was noted; consequently, trim and fill analysis was performed (2 imputed studies) and the results were still insignificant (ES = -0.54; 95% CI: 0.01, −0.26, P < 0.05).
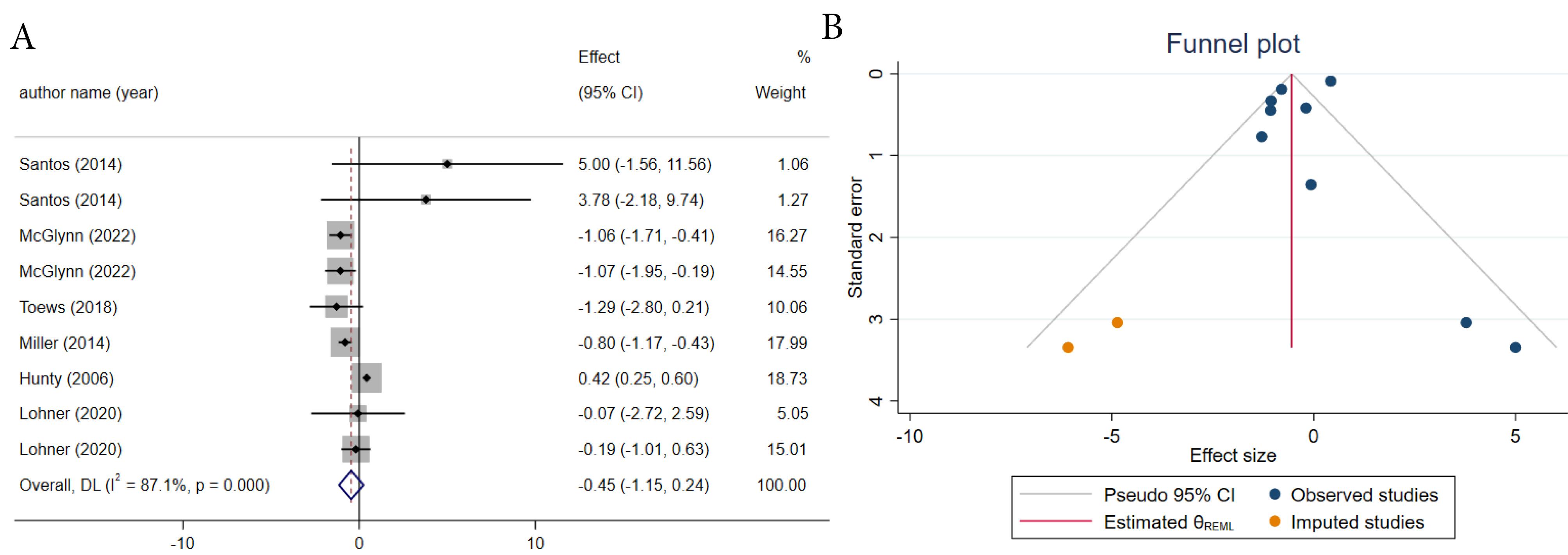
Figure 2.
The Forest plot (A) and trimmed funnel plot (B) of effects of artificial sweeteners on body weight based on weighted mean difference analysis
.
The Forest plot (A) and trimmed funnel plot (B) of effects of artificial sweeteners on body weight based on weighted mean difference analysis
Table 3.
Effect of ASs on BW, BMI, body fat, and energy intake
|
|
Effect size, n
|
ES (95% CI)1
|
P
within2
|
I2
(%)3
|
P
-heterogeneity4
|
|
WMD
|
| AS consumption on weight |
| Overall |
9 |
-0.45 (-1.15, 0.24) |
0.201 |
87.1 |
0.000 |
| Type of sweetener |
| Aspartam |
3 |
1.60 (-1.13, 4.33) |
0.251 |
35.2 |
0.213 |
| Different types |
6 |
-0.81 (-1.09, -0.54) |
0.000 |
00.0 |
0.580 |
| Type of comparator |
| Total |
3 |
-0.80 (-1.88, 0.29) |
0.149 |
41.3 |
0.182 |
| Sucrose |
4 |
-0.09 (-1.33, 1.15) |
0.887 |
0.85 |
0.000 |
| Placebo |
1 |
-0.19 (-1.01, 0.63) |
0.650 |
00.00 |
0.000 |
| Water |
1 |
-1.07 (-1.95, -0.19) |
0.017 |
00.00 |
0.000 |
| Duration |
| ≤ 10 |
2 |
-0.99 (-2.30, 0.32) |
0.137 |
0.43 |
0.000 |
| > 10 |
3 |
-0.18 (-1.10, 0.74) |
0.701 |
94.3 |
0.000 |
| NR |
4 |
-0.79 (-1.80, 0.22) |
0.124 |
47.7 |
0.125 |
| Study number |
| ≤ 5 |
5 |
-0.21 (-1.37, 0.95) |
0.718 |
34.1 |
0.194 |
| > 5 |
4 |
-0.59 (-1.48, 0.31) |
0.199 |
95.4 |
0.000 |
| Study quality |
| Moderate |
6 |
-0.91 (-1.32, -0.49) |
0.000 |
21.5 |
0.272 |
| Critically low |
1 |
0.42 (0.24, 0.60) |
0.000 |
00.0 |
0.000 |
| High |
2 |
-0.18 (-0.96, 0.60) |
0.653 |
00.0 |
0.933 |
| AS consumption on energy intake |
| Overall |
4 |
-0.28 (-1.15, 0.59) |
0.527 |
83.3 |
0.000 |
| Type of comparator |
| Total |
2 |
0.05 (-0.88, 0.98) |
0.913 |
84 |
0.012 |
| Sucrose |
2 |
-0.92 (-1.66, -0.19) |
0.014 |
00.0 |
0.389 |
| Study quality |
| Moderate |
3 |
-0.70 (-1.21, -0.19) |
0.007 |
00.0 |
0.491 |
| Critically low |
1 |
0.47 (0.24, 0.70) |
0.000 |
00.0 |
0.000 |
| AS consumption on BMI |
| Overall |
6 |
-0.28 (-0.40, -0.15) |
0.000 |
00.0 |
0.628 |
| Type of comparator |
| Total |
2 |
-0.25 (-0.41, -0.08) |
0.003 |
00.0 |
0.734 |
| Sucrose |
2 |
-0.36 (-0.60, -0.13) |
0.002 |
00.0 |
0.393 |
| Placebo |
1 |
-0.46 (-0.95, 0.03) |
0.069 |
00.0 |
. |
| Water |
1 |
0.02 (-0.46, 0.50) |
0.936 |
00.0 |
. |
| Study quality |
| Moderate |
5 |
-0.27 (-0.40, -0.15) |
0.000 |
00.0 |
0.492 |
| High |
1 |
-0.37 (-1.10, 0.36) |
0.321 |
00.0 |
. |
| AS consumption on body fat |
| Overall |
3 |
-0.72 (-1.06, -0.37) |
0.000 |
00.0 |
0.392 |
|
SMD
|
| AS consumption on weight |
| Overall |
10 |
-0.38 (-0.56, -0.20) |
0.000 |
61.7 |
0.005 |
| Type of comparator |
| Total |
1 |
-0.40 (-0.58, -0.22) |
0.000 |
00.0 |
. |
| Sucrose |
3 |
-0.69 (-0.99, -0.38) |
0.000 |
50.4 |
0.133 |
| Placebo |
1 |
-0.28 (-0.81, 0.25) |
0.296 |
00.0 |
. |
| Water/nothing |
4 |
-0.24 (-0.47, -0.00) |
0.046 |
00.0 |
0.692 |
| Placebo/nothing |
1 |
-0.06 (-0.27, 0.15) |
0.575 |
00.0 |
. |
| Study quality |
|
|
|
|
|
| Moderate |
4 |
-0.32 (-0.55, -0.09) |
0.007 |
73.5 |
0.010 |
| High |
6 |
-0.45 (-0.75, -0.15) |
0.004 |
54.3 |
0.053 |
| Study number |
|
|
|
|
|
| < 10 |
6 |
-0.19 (-0.33, -0.05) |
0.010 |
00.0 |
0.443 |
| 10 |
4 |
-0.56 (-0.84, -0.27) |
0.000 |
66.5 |
0.030 |
1Obtained from the Random-Effect model. 2Refers to the mean (95% CI). 3Inconsistency, percentage of variation across studies due to heterogeneity. 4Obtained from the Q-test
Effect of ASs on BW in studies reporting SMD
In the meta-analysis of ten included studies which reported SMD, AS significantly reduced BW (ES = -0.38; 95% CI: -0.56, -0.20, P ≤ 0.001) (Figure 3A). A significant between-study heterogeneity was observed (I2 = 61.7 %, P = 0.005) (Figure 3A). The type of comparator, the number of included studies, can be responsible for heterogeneity. In a subgroup analysis of comparator type, AS had a significant BW reduction effect compared to sucrose. Also, both moderate and high-quality studies, and included study numbers of < 10 and 10 indicated a pronounced reduction in BW after AS intervention. The results of the sensitivity analysis revealed that no individual study significantly influenced the overall effect size. Upon visual examination of the funnel plot (Figure 3B), an uneven distribution of studies was observed, prompting the conduct of trim and fill analysis, which imputed one additional study. Despite this, the results remained significant (ES = -0.39; 95% CI: -0.57, −0.21, P < 0.05).
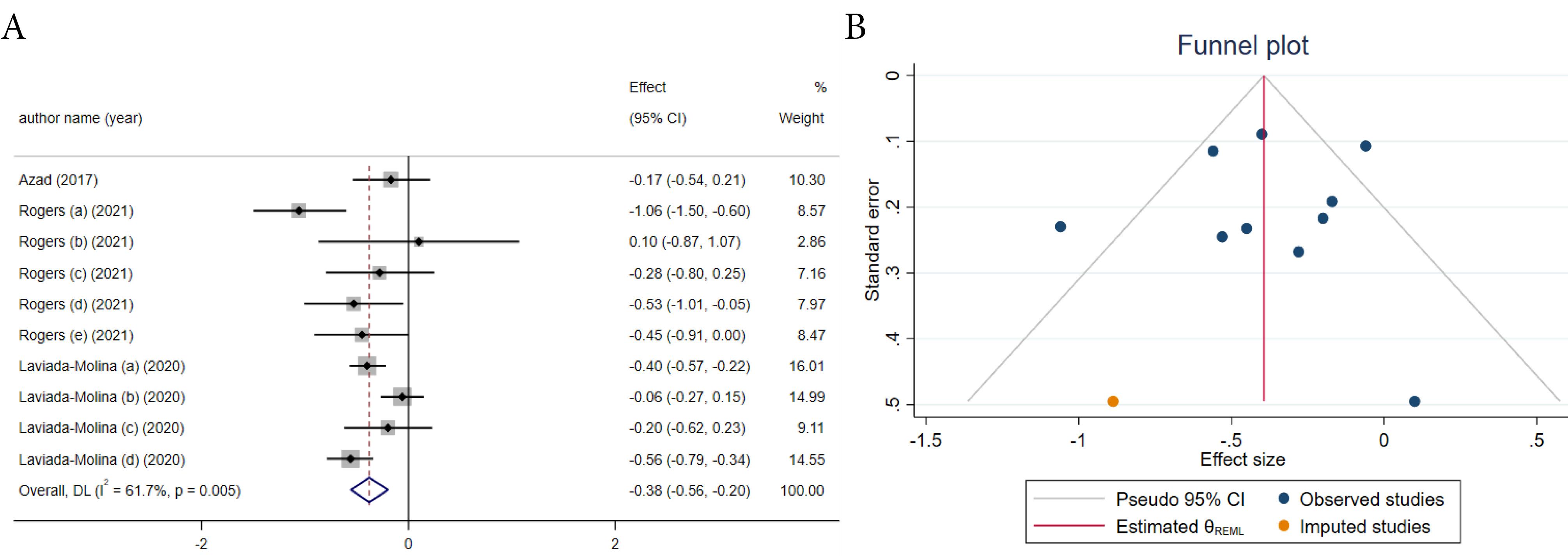
Figure 3.
The Forest plot (A) and trimmed funnel plot (B) of effects of artificial sweeteners on body weight based on standardized mean difference analysis
.
The Forest plot (A) and trimmed funnel plot (B) of effects of artificial sweeteners on body weight based on standardized mean difference analysis
Effect of ASs on energy intake in studies reporting WMD
AS has no significant effect on energy intake in the analysis of four included studies (ES = -0.28; 95% CI: -1.15, 0.59, P = 0.59) (Figure 4). While, in subgroup analysis, energy intake was reduced by AS consumption when compared to sucrose (ES = -0.92; 95% CI: -1.66, -0.19, P = 0.01) (Table 3). A significant between-study heterogeneity was found (I2 = 83.3 %, P ≤ 0.001) (Figure 4A). The study quality and comparator type might be potential sources of heterogeneity. The exclusion of one study25 affected the overall effect size in the sensitivity analysis and the effect of AS in reducing energy intake was significant (ES = -0.69; 95% CI: -1.21, -0.18).
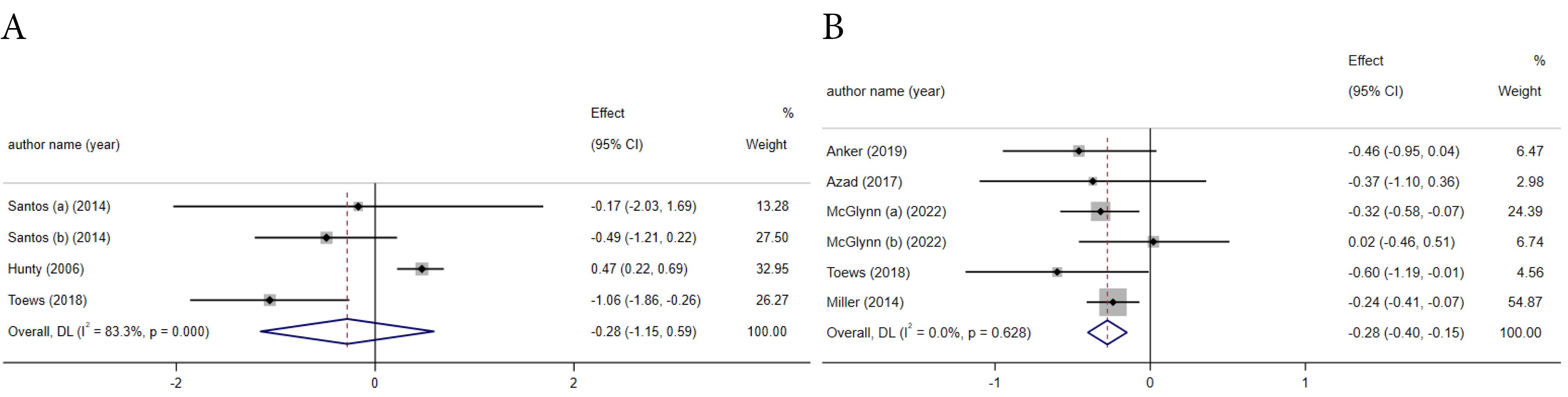
Figure 4.
The effects of artificial sweeteners on energy intake (A) and BMI (B) based on weighted mean difference analysis
.
The effects of artificial sweeteners on energy intake (A) and BMI (B) based on weighted mean difference analysis
Effect of ASs on BMI in studies reporting WMD
The impact of ASs on BMI was examined in six meta-analyses, revealing a significant reduction in BMI attributed to AS consumption (ES = -0.28; 95% CI: -0.40, -0.15, P ≤ 0.001) (Figure 4B). Additionally, the effect of AS in diminishing BMI was more effective when compared to sucrose (Table 3). No between-study heterogeneity was observed. The sensitivity analysis revealed that no individual study had a significant impact on the overall effect size.
Effect of ASs on body fat in studies reporting WMD
AS significantly decreased body fat in a meta-analysis of three included studies (ES = -0.72; 95% CI: -1.06, -0.37, P ≤ 0.001) and between-study heterogeneity was not found (Figure 5A). The results of sensitivity analysis indicated that none of the studies affected the overall effect size.

Figure 5.
The effects of artificial sweeteners on body fat based on weighted mean difference analysis (A) and the risk of overweight and obesity following artificial sweeteners consumption based on umbrella meta-analysis of observational meta-analyses
.
The effects of artificial sweeteners on body fat based on weighted mean difference analysis (A) and the risk of overweight and obesity following artificial sweeteners consumption based on umbrella meta-analysis of observational meta-analyses
Risk of obesity and over weight, and ASs
Three observational studies documented an association between ASs and BW. In contrast to the findings of interventional studies, the meta-analysis of three observational studies indicated that increased consumption of ASs is significantly associated with a heightened risk of obesity and overweight (ES = 1.61; 95% CI: 1.36, 1.87, P ≤ 0.001) (Figure 5B). The results of sensitivity analysis showed that no study affected the overall effect size.
Discussion
The current meta-analysis, which synthesized findings from twelve meta-analyses, investigated the impact of ASs on BW and energy intake. The study participant’s mean age varied from 27 to 66 years. The intervention duration varied between 5 to 48 weeks. The people included in these studies were overweight, obese, diabetic or pre-diabetic, and hypertensive individuals. The number of studies incorporated into the meta-analyses ranged from 2 to 29. The result of this study supported the evidence suggesting that ASs decreased BW in studies reporting SMD, BMI, and body fat in studies reporting WMD. The meta-analysis of observational studies reported that increased consumption of ASs is associated with a significant risk of obesity and overweight.
In our study, the impact of ASs on BW was examined in nine meta-analyses reporting WMD. The combined effect sizes of included studies did not reveal a significant deduction in BW. In a subgroup analysis of comparator type, ASs significantly reduced BW when compared to water. Our result was in accordance with another study which proved that there was a significant reduction in BW on AS consumption compared with water.18 Another study reported that there was no favorable effect of ASs on BW compared with water.26 The selection of studies based on the inclusion criteria differs among each study, which would have contributed to this variation in results.
In our meta-analysis of ten included studies that reported SMD, ASs significantly reduced BW. In a subgroup analysis of comparator type, ASs had a significant BW reduction effect compared to sucrose. This loss of weight might be due to the fact that low-calorie sweeteners could be an alternative to high-calorie drinks and foods. Weight loss was observed in a study using aspartame.25 Following our study results, a greater reduction in BW was observed with ASs compared with sucrose.26 The studies included in our meta-analysis consisted of overweight or obese people, which showed the positive effect of ASs on BW. Moreover, Toews et al indicated a significant reduction in weight in obese or overweight subjects.20
In our study, ASs did not show a significant effect on energy intake in an analysis of four included studies. In the subgroup analysis, energy intake was reduced by AS consumption compared to sucrose. Similar to our results of subgroup analysis, AS consumption reduced food intake for the whole day when compared to sucrose.34 However, a randomized cross-over study found that ASs do not reduce energy intake, compared to sucrose.35 In contrast to our study results, another study declared that aspartame showed a significant decrease in energy intake with all the controls except water.25 Various factors like a deficit of calories, food type, drink type, and duration may be responsible for this contrast. Small fractions of saccharin, rebA, and sucralose have the potential to induce alterations in the colonic microbiota, which in turn could either increase or decrease energy absorption efficiency, thereby impacting BW.36 ASs do not give complete satisfaction to consumers, which may drive them to seek more food intake and thus cause an increase in energy intake or no change in energy intake. Nevertheless, reduced satisfaction does not lead to an increase in energy intake.34 Randomized controlled trials have reported that alternating natural sugars with ASs causes an increase in energy intake.37 However, the intake of energy is still lower with ASs than natural sugars, even after excess energy intake. Therefore, excessive energy intake does not necessarily result in weight gain and may aid in weight maintenance or even weight loss.
The effect of ASs on BMI was reported in six meta-analyses, and ASs were significantly effective in reducing BMI. Additionally, the impact of ASs in reducing BMI was more prominent when compared to sucrose. However, in contrast with our study result, clinical trials have reported that AS administration does not show a significant reduction in BW or BMI.38 Moreover, the non-significant effects of ASs on energy intake suggested that non-caloric properties of ASs would be responsible for boy weight or BMI reduction.39 Majority of the studies incorporated in the meta-analysis took into account lifestyle modifications including diet and physical activity, which would have played a major role in reducing BW and BMI.
ASs significantly decreased body fat in a meta-analysis of three included studies. The same result was obtained in another study, wherein there was a reduction in body fat linked to the consumption of low- and no-calorie sweetened beverages as replacements for sugar-sweetened beverages, resulting in calorie displacement.28
Three observational studies reported an association between ASs and BW. In contrast to the results of intervention studies, in the meta-analysis of three observational studies, increased consumption of ASs is associated with a significant risk of obesity and overweight. Previously conducted reviews15,18 have reported that even though randomized controlled trials have shown that ASs were effective in weight loss, inconsistent results have been obtained from observational studies.
Various meta-analyses of observational studies have evaluated the impact of ASs on BW, yielding conflicting results.18-20 A meta-analysis of randomized controlled trials and prospective cohort studies reported that there was a moderate positive correlation between ASs and BMI but not with BW.15 Another study reported that AS consumption will not result in gaining BW.18 Another meta-analysis proved that ASs resulted in gaining BW.18 Different statistical methods used in different studies and inclusion criteria variability would have been the reason for these varying results. The majority of the included studies for our meta-analysis were conducted on people who were overweight, obese, diabetic or pre-diabetic, and hypertensive. Dietary difference, food-disease interaction, confounding bias and consumer bias may lead to conflicting results.
The findings of our study guide the effective role of ASs on obesity indices and thus as a sugar-reduction strategy. In several countries, there has been a focus mainly on sugar-sweetened beverages,40 since consumption of high sugar may lead to diabetes, obesity, hypertension, and cardiovascular disease.41,42 Previously water was considered a substitute for sugar-sweetened beverages.8 Now based on the evidence from our study, ASs can be used as a substitute for sugar. Especially for individuals who cannot switch to water and who are habited to sugar, ASs would be the best alternative. ASs could be used for maintaining weight loss.43
Strengths and limitation
Evidence was collected and analyzed from both randomized controlled trials and observational studies. The major limitation of umbrella review is the inevitable overlapping. Since most of the studies did not provide information about different kinds of ASs, we could not evaluate them. Since the duration of interventions in a meta-analysis of RCTs varied from 5 to 48 weeks, we could not study the long-term effect of ASs. Moreover, observational studies are more prone to confounding bias.
Conclusion
Evidence from interventional studies supports the benefits of ASs on BW, BMI, and body fat. Contrastingly, evidence from observational studies has proven that augmented consumption of ASs is associated with a significant risk of obesity and overweight which indicates the different effects of long-term and short-term consumption. Future studies should be conducted focusing on the types, dose, and formulations of ASs, to find the overall impact of using ASs as an alternative to sugar. Use of ASs should be used with caution since the long-term benefits and risks were not assessed.
Competing Interests
The authors state that they have no conflicts of interest.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Ethical Approval
Our manuscript is a systematic review that does not involve any experiments or interventions on human, animal, or cell samples. We have conducted a comprehensive literature search and synthesis of existing published data, without performing any direct data collection or experimentation.
Funding
None.
References
- Müller MJ, Geisler C. Defining obesity as a disease. Eur J Clin Nutr 2017; 71(11):1256-8. doi: 10.1038/ejcn.2017.155 [Crossref] [ Google Scholar]
- Powell-Wiley TM, Poirier P, Burke LE, Després JP, Gordon-Larsen P, Lavie CJ. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 2021; 143(21):e984-1010. doi: 10.1161/cir.0000000000000973 [Crossref] [ Google Scholar]
- De Pergola G, Silvestris F. Obesity as a major risk factor for cancer. J Obes 2013; 2013:291546. doi: 10.1155/2013/291546 [Crossref] [ Google Scholar]
- Sarwer DB, Polonsky HM. The psychosocial burden of obesity. Endocrinol Metab Clin North Am 2016; 45(3):677-88. doi: 10.1016/j.ecl.2016.04.016 [Crossref] [ Google Scholar]
- Nagai M, Kuriyama S, Kakizaki M, Ohmori-Matsuda K, Sone T, Hozawa A. Impact of obesity, overweight and underweight on life expectancy and lifetime medical expenditures: the Ohsaki Cohort Study. BMJ Open 2012; 2(3):e000940. doi: 10.1136/bmjopen-2012-000940 [Crossref] [ Google Scholar]
- Okunogbe A, Nugent R, Spencer G, Ralston J, Wilding J. Economic impacts of overweight and obesity: current and future estimates for eight countries. BMJ Glob Health 2021; 6(10):e006351. doi: 10.1136/bmjgh-2021-006351 [Crossref] [ Google Scholar]
- Siervo M, Montagnese C, Mathers JC, Soroka KR, Stephan BC, Wells JC. Sugar consumption and global prevalence of obesity and hypertension: an ecological analysis. Public Health Nutr 2014; 17(3):587-96. doi: 10.1017/s1368980013000141 [Crossref] [ Google Scholar]
- Johnson RK, Appel LJ, Brands M, Howard BV, Lefevre M, Lustig RH. Dietary sugars intake and cardiovascular health: a scientific statement from the American Heart Association. Circulation 2009; 120(11):1011-20. doi: 10.1161/circulationaha.109.192627 [Crossref] [ Google Scholar]
- Buyken AE, Mela DJ, Dussort P, Johnson IT, Macdonald IA, Stowell JD. Dietary carbohydrates: a review of international recommendations and the methods used to derive them. Eur J Clin Nutr 2018; 72(12):1625-43. doi: 10.1038/s41430-017-0035-4 [Crossref] [ Google Scholar]
- Pang MD, Goossens GH, Blaak EE. The impact of artificial sweeteners on body weight control and glucose homeostasis. Front Nutr 2020; 7:598340. doi: 10.3389/fnut.2020.598340 [Crossref] [ Google Scholar]
- Fowler SPG. Low-calorie sweetener use and energy balance: results from experimental studies in animals, and large-scale prospective studies in humans. Physiol Behav 2016; 164(Pt B):517-23. doi: 10.1016/j.physbeh.2016.04.047 [Crossref] [ Google Scholar]
- Swithers SE. Artificial sweeteners produce the counterintuitive effect of inducing metabolic derangements. Trends Endocrinol Metab 2013; 24(9):431-41. doi: 10.1016/j.tem.2013.05.005 [Crossref] [ Google Scholar]
- Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet 2012; 112(5):739-58. doi: 10.1016/j.jand.2012.03.009 [Crossref] [ Google Scholar]
- Pereira MA. Diet beverages and the risk of obesity, diabetes, and cardiovascular disease: a review of the evidence. Nutr Rev 2013; 71(7):433-40. doi: 10.1111/nure.12038 [Crossref] [ Google Scholar]
- Miller PE, Perez V. Low-calorie sweeteners and body weight and composition: a meta-analysis of randomized controlled trials and prospective cohort studies. Am J Clin Nutr 2014; 100(3):765-77. doi: 10.3945/ajcn.113.082826 [Crossref] [ Google Scholar]
- de Koning L, Malik VS, Rimm EB, Willett WC, Hu FB. Sugar-sweetened and artificially sweetened beverage consumption and risk of type 2 diabetes in men. Am J Clin Nutr 2011; 93(6):1321-7. doi: 10.3945/ajcn.110.007922 [Crossref] [ Google Scholar]
- Hinkle SN, Rawal S, Bjerregaard AA, Halldorsson TI, Li M, Ley SH. A prospective study of artificially sweetened beverage intake and cardiometabolic health among women at high risk. Am J Clin Nutr 2019; 110(1):221-32. doi: 10.1093/ajcn/nqz094 [Crossref] [ Google Scholar]
- Rogers PJ, Hogenkamp PS, de Graaf C, Higgs S, Lluch A, Ness AR. Does low-energy sweetener consumption affect energy intake and body weight? A systematic review, including meta-analyses, of the evidence from human and animal studies. Int J Obes (Lond) 2016; 40(3):381-94. doi: 10.1038/ijo.2015.177 [Crossref] [ Google Scholar]
- Azad MB, Abou-Setta AM, Chauhan BF, Rabbani R, Lys J, Copstein L. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. Cmaj 2017; 189(28):E929-e39. doi: 10.1503/cmaj.161390 [Crossref] [ Google Scholar]
- Toews I, Lohner S, Küllenberg de Gaudry D, Sommer H, Meerpohl JJ. Association between intake of non-sugar sweeteners and health outcomes: systematic review and meta-analyses of randomised and non-randomised controlled trials and observational studies. BMJ 2019; 364:k4718. doi: 10.1136/bmj.k4718 [Crossref] [ Google Scholar]
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev 2015; 4(1):1. doi: 10.1186/2046-4053-4-1 [Crossref] [ Google Scholar]
- Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017; 358:j4008. doi: 10.1136/bmj.j4008 [Crossref] [ Google Scholar]
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions: John Wiley & Sons; 2019.
- Bundgaard Anker CC, Rafiq S, Jeppesen PB. Effect of steviol glycosides on human health with emphasis on type 2 diabetic biomarkers: a systematic review and meta-analysis of randomized controlled trials. Nutrients 2019; 11(9):1965. doi: 10.3390/nu11091965 [Crossref] [ Google Scholar]
- de la Hunty A, Gibson S, Ashwell M. A review of the effectiveness of aspartame in helping with weight control. Nutr Bull 2006; 31(2):115-28. doi: 10.1111/j.1467-3010.2006.00564.x [Crossref] [ Google Scholar]
- Laviada-Molina H, Molina-Segui F, Pérez-Gaxiola G, Cuello-García C, Arjona-Villicaña R, Espinosa-Marrón A. Effects of nonnutritive sweeteners on body weight and BMI in diverse clinical contexts: systematic review and meta-analysis. Obes Rev 2020; 21(7):e13020. doi: 10.1111/obr.13020 [Crossref] [ Google Scholar]
- Lohner S, Kuellenberg de Gaudry D, Toews I, Ferenci T, Meerpohl JJ. Non-nutritive sweeteners for diabetes mellitus. Cochrane Database Syst Rev 2020; 5(5):CD012885. doi: 10.1002/14651858.CD012885.pub2 [Crossref] [ Google Scholar]
- McGlynn ND, Khan TA, Wang L, Zhang R, Chiavaroli L, Au-Yeung F. Association of low- and no-calorie sweetened beverages as a replacement for sugar-sweetened beverages with body weight and cardiometabolic risk: a systematic review and meta-analysis. JAMA Netw Open 2022; 5(3):e222092. doi: 10.1001/jamanetworkopen.2022.2092 [Crossref] [ Google Scholar]
- Qin P, Li Q, Zhao Y, Chen Q, Sun X, Liu Y. Sugar and artificially sweetened beverages and risk of obesity, type 2 diabetes mellitus, hypertension, and all-cause mortality: a dose-response meta-analysis of prospective cohort studies. Eur J Epidemiol 2020; 35(7):655-71. doi: 10.1007/s10654-020-00655-y [Crossref] [ Google Scholar]
- Rogers PJ, Appleton KM. The effects of low-calorie sweeteners on energy intake and body weight: a systematic review and meta-analyses of sustained intervention studies. Int J Obes (Lond) 2021; 45(3):464-78. doi: 10.1038/s41366-020-00704-2 [Crossref] [ Google Scholar]
- Ruanpeng D, Thongprayoon C, Cheungpasitporn W, Harindhanavudhi T. Sugar and artificially sweetened beverages linked to obesity: a systematic review and meta-analysis. QJM 2017; 110(8):513-20. doi: 10.1093/qjmed/hcx068 [Crossref] [ Google Scholar]
- Santos NC, de Araujo LM, De Luca Canto G, Guerra EN, Coelho MS, de Fatima Borin M. Metabolic effects of aspartame in adulthood: a systematic review and meta-analysis of randomized clinical trials. Crit Rev Food Sci Nutr 2018; 58(12):2068-81. doi: 10.1080/10408398.2017.1304358 [Crossref] [ Google Scholar]
- Reid AE, Chauhan BF, Rabbani R, Lys J, Copstein L, Mann A. Early exposure to nonnutritive sweeteners and long-term metabolic health: a systematic review. Pediatrics 2016; 137(3):e20153603. doi: 10.1542/peds.2015-3603 [Crossref] [ Google Scholar]
- Anton SD, Martin CK, Han H, Coulon S, Cefalu WT, Geiselman P. Effects of stevia, aspartame, and sucrose on food intake, satiety, and postprandial glucose and insulin levels. Appetite 2010; 55(1):37-43. doi: 10.1016/j.appet.2010.03.009 [Crossref] [ Google Scholar]
- Tey SL, Salleh NB, Henry J, Forde CG. Effects of aspartame-, monk fruit-, stevia- and sucrose-sweetened beverages on postprandial glucose, insulin and energy intake. Int J Obes (Lond) 2017; 41(3):450-7. doi: 10.1038/ijo.2016.225 [Crossref] [ Google Scholar]
- Magnuson BA, Carakostas MC, Moore NH, Poulos SP, Renwick AG. Biological fate of low-calorie sweeteners. Nutr Rev 2016; 74(11):670-89. doi: 10.1093/nutrit/nuw032 [Crossref] [ Google Scholar]
- Naismith DJ, Rhodes C. Adjustment in energy intake following the covert removal of sugar from the diet. J Hum Nutr Diet 1995; 8(3):167-75. doi: 10.1111/j.1365-277X.1995.tb00309.x [Crossref] [ Google Scholar]
- Maki KC, Curry LL, Reeves MS, Toth PD, McKenney JM, Farmer MV. Chronic consumption of rebaudioside A, a steviol glycoside, in men and women with type 2 diabetes mellitus. Food Chem Toxicol 2008; 46 Suppl 7:S47-53. doi: 10.1016/j.fct.2008.05.007 [Crossref] [ Google Scholar]
- Gardner C, Wylie-Rosett J, Gidding SS, Steffen LM, Johnson RK, Reader D. Nonnutritive sweeteners: current use and health perspectives: a scientific statement from the American Heart Association and the American Diabetes Association. Circulation 2012; 126(4):509-19. doi: 10.1161/CIR.0b013e31825c42ee [Crossref] [ Google Scholar]
- Malik VS, Hu FB. Sugar-sweetened beverages and cardiometabolic health: an update of the evidence. Nutrients 2019; 11(8):1840. doi: 10.3390/nu11081840 [Crossref] [ Google Scholar]
- Imamura F, O’Connor L, Ye Z, Mursu J, Hayashino Y, Bhupathiraju SN. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: systematic review, meta-analysis, and estimation of population attributable fraction. BMJ 2015; 351:h3576. doi: 10.1136/bmj.h3576 [Crossref] [ Google Scholar]
- Xi B, Huang Y, Reilly KH, Li S, Zheng R, Barrio-Lopez MT. Sugar-sweetened beverages and risk of hypertension and CVD: a dose-response meta-analysis. Br J Nutr 2015; 113(5):709-17. doi: 10.1017/s0007114514004383 [Crossref] [ Google Scholar]
- Catenacci VA, Pan Z, Thomas JG, Ogden LG, Roberts SA, Wyatt HR. Low/no calorie sweetened beverage consumption in the National Weight Control Registry. Obesity (Silver Spring) 2014; 22(10):2244-51. doi: 10.1002/oby.20834 [Crossref] [ Google Scholar]